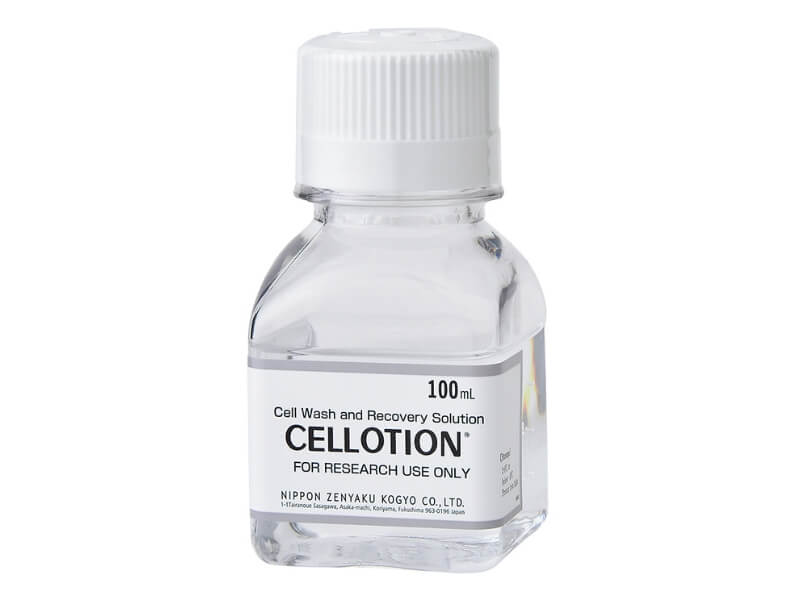
CELLOTION®
Increase the yield of cell recovery
Product Basics
CELLOTION® is a chemically defined cell washing solution that helps to increase the yield of cell recovery. The unique formulation free of serum, protein, and sugars, is ideal for cells, tissues, and organs.
Key Features
- Significantly increase cell yield after washing procedures while maintaining cell viability
- Free of serum and animal-derived components
- Chemically defined solution
- Simple protocol
Technical Information
Why choose CELLOTION® Cell wash and recovery solution
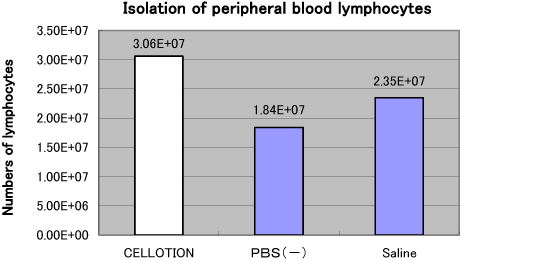
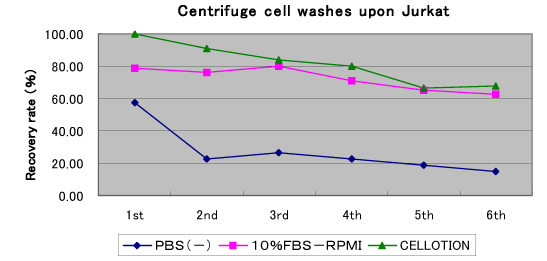
A row of cell washes for each cell isolation or post-thawing manipulation may cause plenty of cell loss, particularly if the process conducted only with serum and protein-free medium. Imagine that as much as 40% of your valuable cells may go down into the drain unawarely every time after cell wash, CELLOTION® ensures you with better yields of cells.
Figure 1. Comparative Analysis of CELLOTION® Cell Wash and Recovery Solution vs. General-use Cell Wash Technologies
Figure 2. Comparative Analysis (multiple washing procedures) of CELLOTION Cell Wash and Recovery Solution vs. General-use Cell Wash Technologies
Application
- Stem cell isolation from a variety of sources
- Leukocyte isolation from peripheral blood
- Post-thawing cell wash
- General cell wash
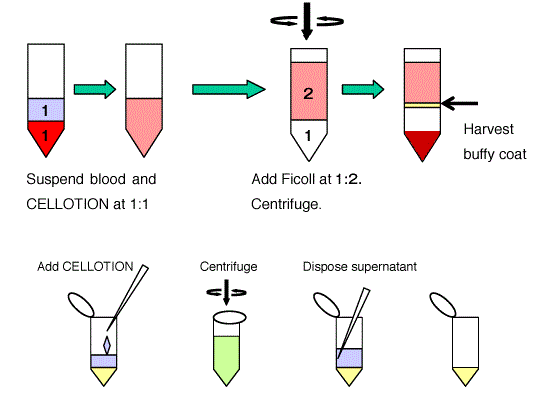
Protocol
- Gently dilute the peripheral blood or umbilical cord blood (UCB) with CELLOTION® Cell Wash and Recovery Solution 1:1.
- In a new tube add Ficoll then the blood prepared in step 1. Blood should be added gently, layering on top of Ficoll. Do not let it mix. The amount of Ficoll to blood should be 1:2.
- Centrifuge the tube for 30 minutes at 1,800 rpm, at room temperature.
- Collect the buffy coat (monocytes layer) carefully with a sterile pipette tip. Avoid pipetting Ficoll as much as possible.
- Gently dilute the cells with CELLOTION®, then wash 2 – 3 times in CELLOTION®.
- Count the number of cells.
Specification
- Manufactured By : Zenogen Pharma Co., Ltd
- Size : 100mL (SKU: 11918)
- Storage and Stability : Store at 2 to 8℃
- Expiration date : 3 years after the date of manufacture
- Disclaimer: For research purposes only
Pricing
CELLOTION®
- For washing cells
- Higher recovery
- Animal component free・Protein free
- SKU: 11918
- Size: 100mL
- Price: $60.00
References & Literature
Yamashiro, C., Sasaki, K., Yokobayashi, S., Kojima, Y. & Saitou, M. Generation of human oogonia from induced pluripotent stem cells in culture. Nature Protocols 15, 1560–1583 (2020) doi: 10.1038/s41596-020-0297-5.
Murase, Y. et al. Long-term expansion with germline potential of human primordial germ cell-like cells in vitro. The EMBO Journal 39, e104929 (2020) doi: 10.15252/embj.2020104929.
Hwang, Y. S. et al. Reconstitution of prospermatogonial specification in vitro from human induced pluripotent stem cells. Nature Communications 11, 5656 (2020) doi: 10.1038/s41467-020-19350-3.
Yamamoto, S. et al. Quantitative PCR methodology with a volume-based unit for the sophisticated cellular kinetic evaluation of chimeric antigen receptor T cells. Scientific Reports 10, 17884 (2020) doi: 10.1038/s41598-020-74927-8.
Related Products
FOR RESEARCH USE ONLY, NOT FOR USE IN DIAGNOSTIC PROCEDURES