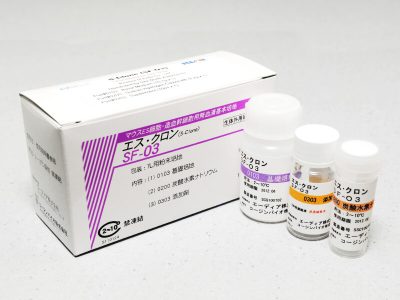
S-Clone™ SF-O3
A serum-free medium for mouse hematopoietic stem cells
Product Basics
S-Clone SF-03 is a completely serum-free medium for use in research of mouse hematopoietic stem cells. The product was developed based upon the results of research by the Nishikawa Laboratory, part of the Department of Molecular Genetics at the Graduate School of Medicine, Kyoto University. It is used for molecular studies of proliferation and maintenance mechanisms of mouse hematopoietic stem cells and has been reported effective in the same.
The medium is serum-free, eliminating interference by unknown factors in serum, and it contains extremely limited protein components, allowing the action of cytokines, etc. to be clearly identified. Being composed of known ingredients, the product affords superior constancy in experimental conditions.
Key Features
- Mouse Hematopoietic Stem Cells
- Serum Free
Technical Information
Formulation
1. Basal medium (powder)
Constituted from a composite of slightly modified RPMI1640, Dulbecco’s MEM, and Ham’s F-12.
2. Sodium hydrogen carbonate (powder)
3. Supplement (liquid)
HEPES buffer containing hormone growth factors (insulin, transferrin, sodium selenite and ethanolamine). Protein components other than insulin and transferrin have not been added.
Directions for Use
Preparing the Medium
1. Add 1 bottle of the basal medium (10.7 g) to approximately 950 mL of purified water and mix well to dissolve.
2. To the solution above, add 1 vial (2.2 g) of sodium hydrogen carbonate and dissolve, and further dilute to a total 1,000 mL.
3. Sterilize by filtering through a low adsorption membrane filter of pore size 0.2-0.22 um (Toyo Roshi Kaisha Ltd., Cat. No. C020A047A or Millipore Corporation, Cat. No. GVWP04700, etc.).
4. Aseptically add 1 vial of the supplement (10 mL) to the filtered 1,000 mL of medium.
5. Seal and store refrigerated (2-10C).

Examples of Usage
1. Method for cultivation of B precursor cells on a mouse stromal cell line ST2 layer using SF-O3 (Add IL-7: 10 U/mL, 2- mercaptoethanol: 100 umol/L).
1) Seed the ST2 cells at a density of 1.5 x 10^5 cells/well to make the feeder layer.
2) Seed the mouse bone marrow cells at a density of 2 x 10^5 cells/well on the ST2 feeder layer and continue to incubate, replacing half of the medium every 4-5 days. Well used: Falcon PrimariaCultureware 6-Well Plate (3846)
2. Method for cultivation of hematopoietic precursor cells in a mouse stromal cell line PA6 layer using SF-O3 (Add bFGF or EGF: 5-10 ng/mL, 2- mercaptoethanol: 100 umol/L).
1) Seed the PA6 cells at a density of 2 x 10^5 cells/well to make the feeder layer.
2) Seed the mouse bone marrow cells at a density of 2 x 10^5 cells/well on the PA6 feeder layer and continue to incubate, replacing half of the medium every 4-5 days. Well used: Falcon PrimariaCultureware 6-Well Plate (3846)
Precautions for Use and Handling
Specification
- Storage and Shelf Life:
- Store at 2-10C, avoid freezing.
- The shelf life is 1 year following manufacture (expiration date is indicated on the outer box)
- Package Contents:
- Basal medium: 1 L-equivalent x 1 bottle
- Sodium hydrogen carbonate: 2.2 g x 1 vial
- Supplement: 10 mL x 1 unit vial
- Manufactured by:
Sekisui Medical Co., LTD
Urban Net Nihonbashi 2-chome Building
2-1-3, Nihonbashi, Chuo-ku, Tokyo, Japan 103-0027
Pricing
S-Clone™ SF-O3
- A serum-free medium for use in research of hematopoietic stem cells
- SKU: 1303
- Price: $439.00
References & Literature
- Nishikawa, S. et al. Stromal cell-dependent bone marrow culture with a nearly protein-free defined medium. Immunology Letters 40, 163–169 (1994) doi: 10.1016/0165-2478(94)90188-0.
- Yasunaga, M., Adachi, S., Itoh, N. & Nishikawa, S.-I. Making the in-vitro model closer to actual B lymphopoiesis in the bone marrow. Seminars in Immunology 7, 185–196 (1995) doi: 10.1016/1044-5323(95)90046-2.
- Yasunaga, M., Wang, F., Kunisada, T., Nishikawa, S. & Nishikawa, S. Cell cycle control of c-kit+IL-7R+ B precursor cells by two distinct signals derived from IL-7 receptor and c-kit in a fully defined medium. Journal of Experimental Medicine 182, 315–323 (1995) doi: 10.1084/jem.182.2.315.
- Sase, H., Watabe, T., Kawasaki, K., Miyazono, K. & Miyazawa, K. VEGFR2-PLCγ1 axis is essential for endothelial specification of VEGFR2+ vascular progenitor cells. Journal of Cell Science 122, 3303–3311 (2009) doi: 10.1242/jcs.049908.
- Yasunaga, M. et al. Involvement of Fyn tyrosine kinase in progression of cytokinesis of B lymphocyte progenitor. J Cell Biol 132, 91–99 (1996) doi: 10.1083/jcb.132.1.91.
- Yasunaga, M. et al. Induction and monitoring of definitive and visceral endoderm differentiation of mouse ES cells. Nature Biotechnology 23, 1542–1550 (2005) doi: 10.1038/nbt1167.
- Sarrazin, S. et al. MafB Restricts M-CSF-Dependent Myeloid Commitment Divisions of Hematopoietic Stem Cells. Cell 138, 300–313 (2009) doi: 10.1016/j.cell.2009.04.057.
- Nakajima, H. et al. TIMP-3 recruits quiescent hematopoietic stem cells into active cell cycle and expands multipotent progenitor pool. Blood 116, 4474–4482 (2010) doi: 10.1182/blood-2010-01-266528.
- Jakt, L. M., Moriwaki, S. & Nishikawa, S. A continuum of transcriptional identities visualized by combinatorial fluorescent in situ hybridization. Development 140, 216–225 (2013) doi: 10.1242/dev.086975.
- Feng, J. et al. Clonal lineage tracing reveals shared origin of conventional and plasmacytoid dendritic cells. Immunity 55, 405-422.e11 (2022) doi: 10.1016/j.immuni.2022.01.016.
- Omatsu, Y. et al. Runx1 and Runx2 inhibit fibrotic conversion of cellular niches for hematopoietic stem cells. Nature Communications 13, 2654 (2022) doi: 10.1038/s41467-022-30266-y.
- Jin, X. et al. Epigenetic downregulation of Socs2 contributes to mutant N-Ras-mediated hematopoietic dysregulation. Disease Models & Mechanisms 15, dmm049088 (2022) doi: 10.1242/dmm.049088.
- Sorimachi, Y. et al. p38α plays differential roles in hematopoietic stem cell activity dependent on aging contexts. Journal of Biological Chemistry 296, (2021) doi: 10.1016/j.jbc.2021.100563.
- Li, K. et al. Interrogation of enhancer function by enhancer-targeting CRISPR epigenetic editing. Nature Communications 11, 485 (2020) doi: 10.1038/s41467-020-14362-5.
Other Documents
FOR RESEARCH USE ONLY, NOT FOR USE IN DIAGNOSTIC PROCEDURES