iMatrix 511 Series
Maintenance and expansion of pluripotent stem cells (PSCs) & mesenchymal stem cells (MSCs)
Product Basics
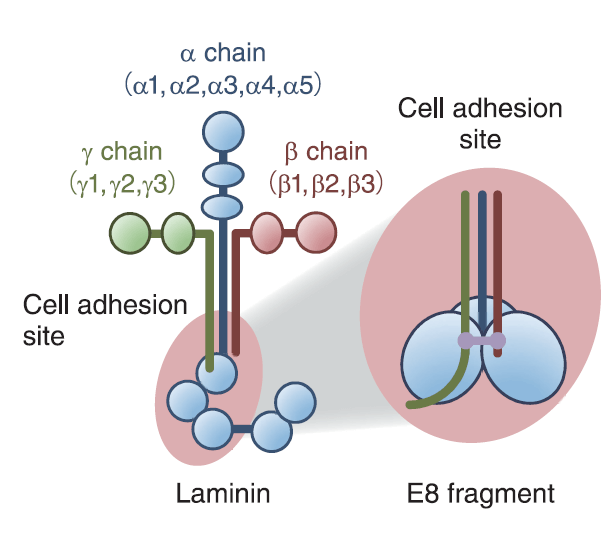
The iMatrix-511 is an innovative cell culture matrix that is compatible with a wide variety of cell types. It is exceptionally well-suited for pluripotent and mesenchymal stem cells. This product consists of recombinant Laminin-511 E8 protein fragments, which allow ES/iPS cells and MSCs to be maintained in xeno-free culture conditions. It also enables the passaging of single cells and provides greater adhesion than full-length Laminin, Vitronectin, or Matrigel® (Miyazaki et al., Nature Commun. 3; 1236 (2012)).
Key Features
*Available in three grades, including GMP, to support a smooth transition from R&D to clinical stages
Advantages vs. Matrigel®
- Defined xeno-free culture
- Expansion efficiency is ~200x higher
- Work is simplified for reduced labor and human error
- Cost: $1–2 per well (iMatrix-511) vs. $3–4 per well (Matrigel®)
Greater Cell Adhesion & Proliferation
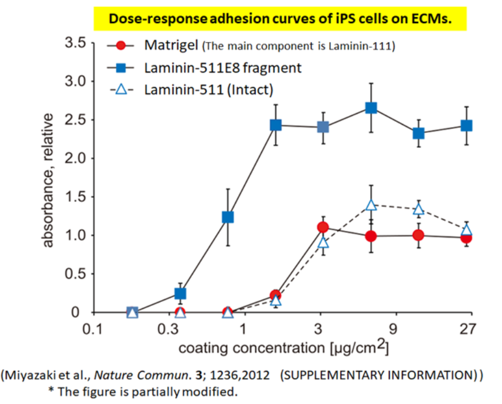
Human iPS cells exhibit stronger adhesion to the Laminin-511 E8 fragment than to the “Laminin-511 (Intact).”
The adhesion strength of the Laminin-511 E8 fragment and other cell culture substrates to human iPS cells was compared. Human iPS cells adhered more strongly to the Laminin-511 E8 fragment than to “Laminin-511 (Intact).”
Absorbance (OD570) represents the relative number of attached cells and was normalized to the maximum response observed on Matrigel®, which was arbitrarily set to 1.
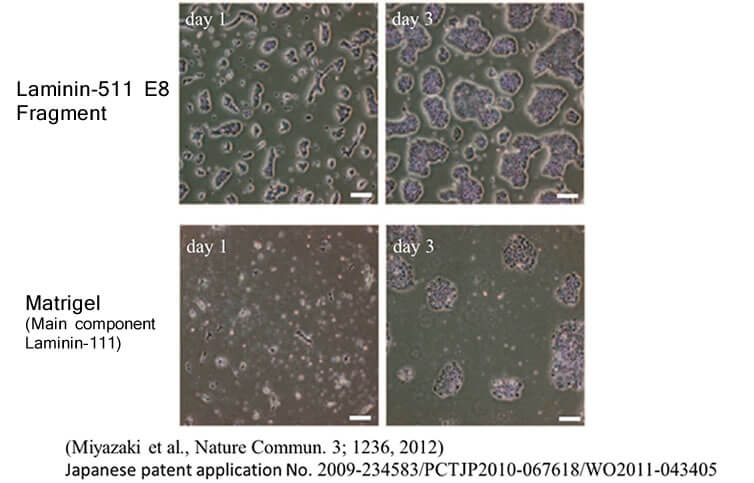
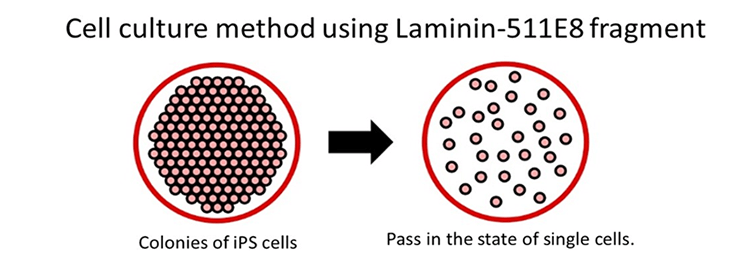
In the same study mentioned above, human ES cells were passaged on Laminin-511 E8 fragment and other cell culture substrates. When human ES cell colonies were dispersed individually at the time of passage, it was observed that single cells adhered rapidly to Laminin-511 E8 and proliferated immediately.
LM-E8s are more effective for single-cell culture than Matrigel®, fibronectin, or vitronectin
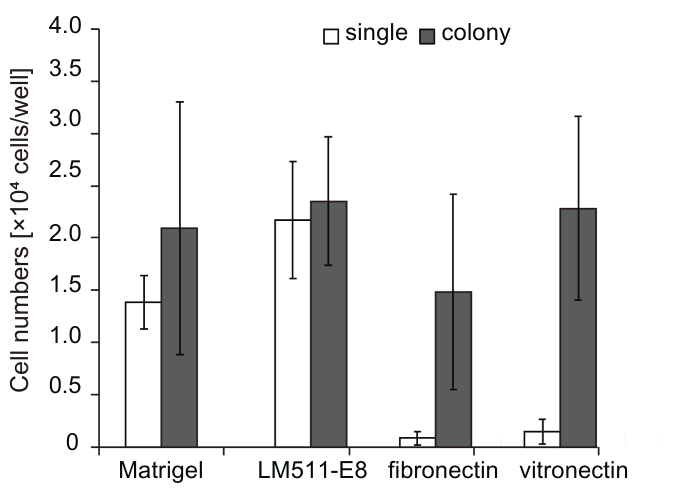
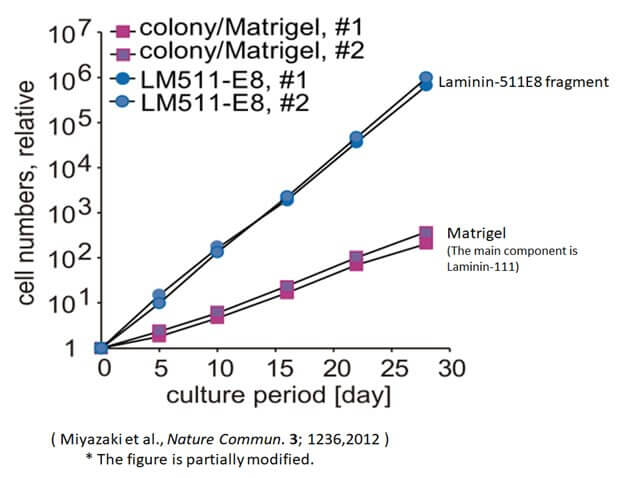
Culture more ES/iPS cells using Laminin-511 E8 fragment
The graph above compares the numbers of ES/iPS cells cultured by the conventional method (colony) for 30 days with those cultured using the Laminin-511 E8 fragment. The results confirm that the cell count increased by at least 200 times when the Laminin-511 E8 fragment was employed.
Technical Information
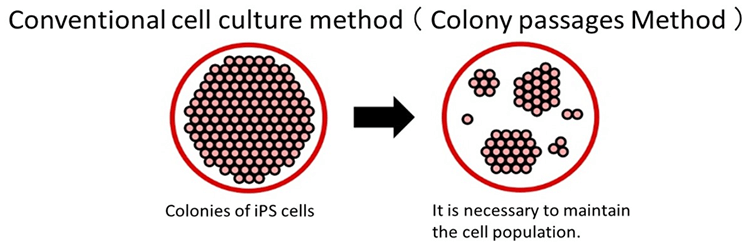
Single-Cell Passaging Ready
With conventional methods, ES/iPS cells perish when dispersed into single cells, necessitating the maintenance of a cell colony during passaging.
Using the Laminin-511 E8 fragment as a cell culture matrix enables the culturing of single ES/iPS cells, which in turn reduces the time required to optimize culture methods and drastically enhances the efficiency of cell cultures.
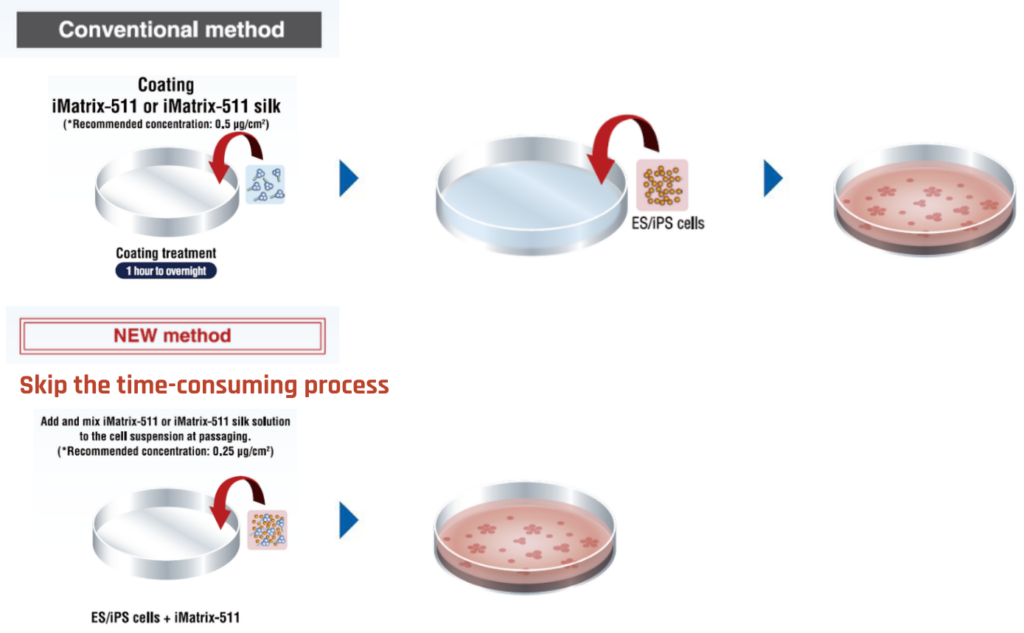
Pre-Mix Method (Without Pre-Coating) For Higher Efficiency
Advantages
- Requires half the amount of iMatrix protein needed compared to pre-coating method
- Lower preparation time (no incubation needed)
- No need for spare pre-coated plates
- Prevents uneven growth due to irregular coating
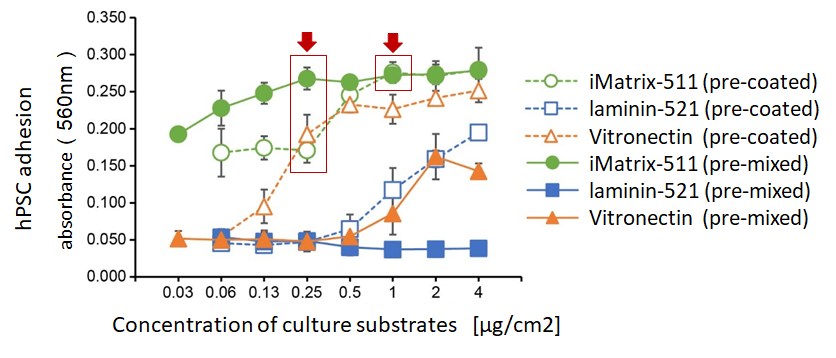
Comparison of iMatrix-511, laminin-521, and recombinant human vitronectin in supporting cell adhesion using pre-coat versus pre-mix methods. The pre-mix method reduces the amount of substrate needed for maximum adhesion of hPSCs, while the iMatrix-511 pre-coating method still supports robust cell growth even with smaller amounts of substrate.
Scraper-Free Cell Detachment Using iMatrix

Easy iMatrix
Ready-to-use Easy iMatrix‑511 provides a reliable Laminin‑511 E8 coating for efficient feeder-free cell culture workflows.
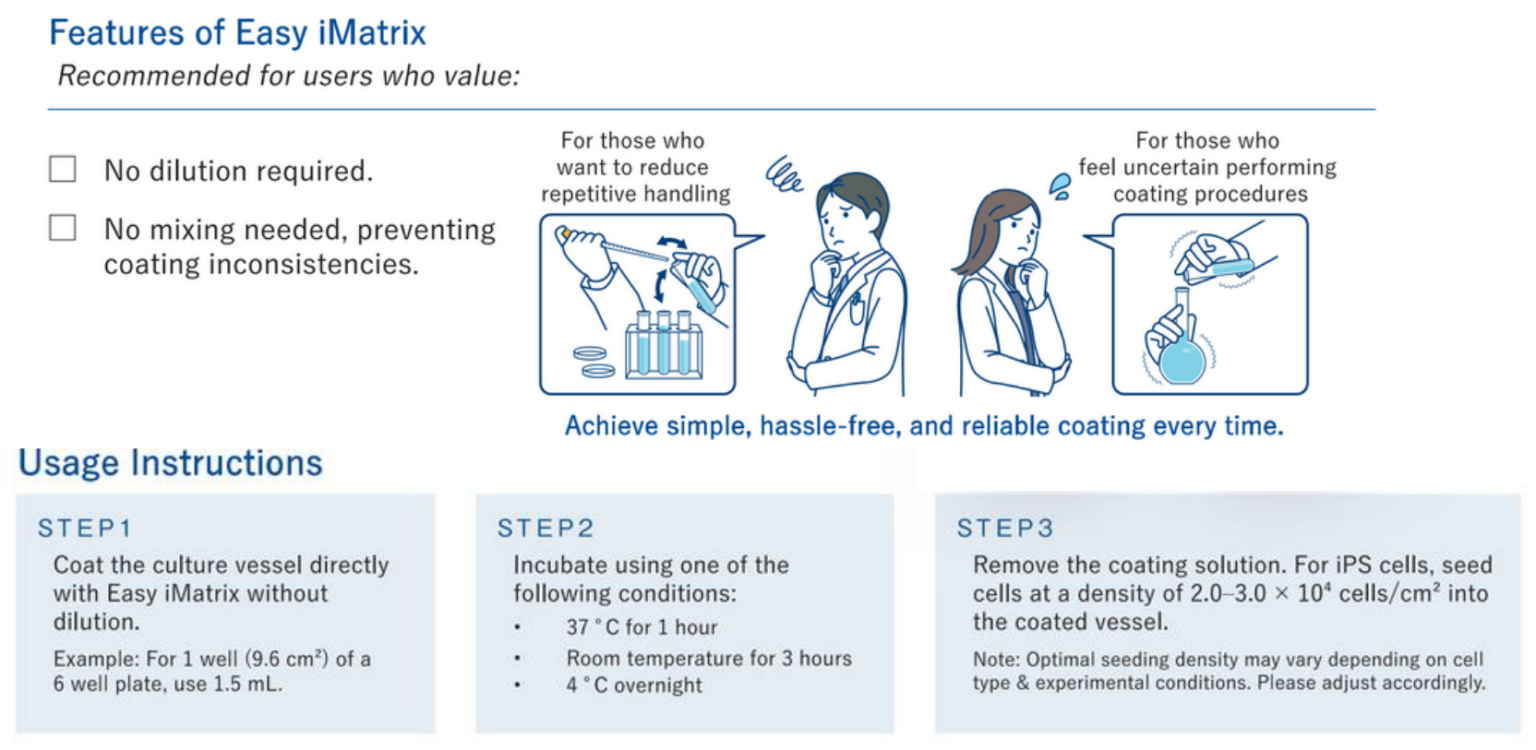
- Ready-to-use: eliminates dilution or mixing
- Pre-diluted at a concentration of 1.6ug/mL
- Simplifies workflows and reduces hands-on preparation
- Ensures consistent, reproducible coating results
Specification
- Format: Liquid Solution (175 ug/vial @0.5 mg/mL), Solvent: PBS (-)
- Storage temperature: 2-15 °C
- Manufactured by : Nippi, Inc. 1-1-1 Senju-Midoricho, Adachi-ku, Tokyo, Japan
- Available in 3 grades:
- iMatrix-511: expressed with CHO-S Cell
- iMatrix-511 SILK: cost-effective version expressed with transgenic silkworm cocoon
- iMatrix-511MG: Clinical-grade (GMP) [Please inquire for more details]
Pricing
iMatrix-511
- Expressed by CHO-S Cell
- Liquid Solution (175 ug/vial @0.5 mg/mL), Solvent: PBS (-)
- 175ug coats roughly 2-6 six well plates via conventional method
- SKU: N-892013
- Price: $80.00
- Incl. US (Continental) shipping fee
- * Only one purchase per lab *
350ug size (175ug x 2 tubes)
- SKU: N-892011
- Price:
$309.00→ $216.00
1050ug size (175ug x 6 tubes)
- SKU: N-892012
- Price:
$725.00→ $565.00
iMatrix-511 Silk
- Expressed by transgenic silkworm cocoon
- Cost-effective alternative to the standard iMatrix-511
- Liquid Solution (175 ug/vial @0.5 mg/mL), Solvent: PBS (-)
- 175ug coats roughly 2-6 six well plates via conventional method
- SKU: N-892022
- Price: $80.00
- Incl. US (Continental) shipping fee
- * Only one purchase per lab *
Easy iMatrix-511
- Pre-diluted at a concentration of 1.6ug/mL
- Ready to Use
- 100mL = roughly 16 6-well plate (amount to coat well surface = 0.25ug/cm²)
Easy iMatrix-511 (CHO-S Cell)
- SKU: N-892019
- Price: $80.00
- Incl. US (Continental) shipping fee
- * Only one purchase per lab *
- SKU: N-892018
- Price: $380.00
Easy iMatrix-511 Silk (Transgenic silkworm cocoon)
- SKU: N-892025
- Price: $80.00
- Incl. US (Continental) shipping fee
- * Only one purchase per lab *
- SKU: N-892024
- Price: $330.00
References & Literature
- Taniguchi, Y. et al. The C-terminal region of laminin beta chains modulates the integrin binding affinities of laminins. J Biol Chem 284, 7820–7831 (2009) doi: 10.1074/jbc.M809332200.
- Miyazaki, T. et al. Laminin E8 fragments support efficient adhesion and expansion of dissociated human pluripotent stem cells. Nature Communications 3, 1236 (2012) doi: 10.1038/ncomms2231.
Imashiro, C. et al. Perfusable System Using Porous Collagen Gel Scaffold Actively Provides Fresh Culture Media to a Cultured 3D Tissue. International Journal of Molecular Sciences 22, 6780 (2021) doi: 10.3390/ijms22136780.
Ito, T. et al. The therapeutic potential of multiclonal tumoricidal T cells derived from tumor infiltrating lymphocyte-derived iPS cells. Communications Biology 4, 694 (2021) doi: 10.1038/s42003-021-02195-x.
Hirano, S. et al. Expansion Culture of Hair Follicle Stem Cells through Uniform Aggregation in Microwell Array Devices. ACS Biomater. Sci. Eng. 9, 1510–1519 (2023) doi: 10.1021/acsbiomaterials.2c01141.
Tanaka, K. et al. Robust and reproducible human intestinal organoid-derived monolayer model for analyzing drug absorption. Scientific Reports 15, 11403 (2025) doi: 10.1038/s41598-025-95823-z.
Akiyama, T., Sato, S., Chikazawa-Nohtomi, N., Soma, A., Kimura, H., Wakabayashi, S., Ko, S. B., Ko, M. S.. Efficient differentiation of human pluripotent stem cells into skeletal muscle cells by combining RNA-based MYOD1-expression and POU5F1-silencing. Scientific Reports, 8(1), 1189, (2018)
Ayabe, H., Anada, T., Kamoya, T., Sato, T., Kimura, M., Yoshizawa, E., Kikuchi, Shunyuu., Ueno, Yasuharu., Sekine, keisuke., J. Gray Camp., Treutlein, B., Ferguson, Autumn., Suzuki, Osamu., Takede, Takanori.. Optimal Hypoxia Regulates Human iPSC-Derived Liver Bud Differentiation through Intercellular TGFB Signaling. Stem Cell Reports, 11, 1-11, (2018)
Ishida, K., Xu, H., Sasakawa, N., Lung, M. S. Y., Kudryashev, J. A., Gee, P., & Hotta, A.. Site-specific randomization of the endogenous genome by a regulatable CRISPR-Cas9 piggyBac system in human cells. Scientific Reports, 8(1), 310, (2018)
Ishii, K., Sakurai, H., Suzuki, N., Mabuchi, Y., Sekiya, I., Sekiguchi, K., Akazawa, C.. Recapitulation of Extracellular LAMININ Environment Maintains Stemness of Satellite Cells In Vitro. Stem Cell Reports, 10, 1-15, (2018)
Kagihiro, M., Fukumori, K., Aoki, T., Ungkulpasvich, U., Mizutani, M., Viravaidya-Pasuwat, K., & Kino-oka, M.. Kinetic analysis of cell decay during the filling process: Application to lot size determination in manufacturing systems for human induced pluripotent and mesenchymal stem cells. Biochemical Engineering Journal, 131, 31-38, (2018)
Kim, S. I., Matsumoto, T., Kagawa, H., Nakamura, M., Hirohata, R., Ueno, A., Ohishi, M., Sakuma, T., Soga, T., Yamamoto, T., Woltjen, K.. Microhomology-assisted scarless genome editing in human iPSCs. Nature Communications, 9(1), 939, (2018)
Li, L., Roh, J. H., Chang, E. H., Lee, Y., Lee, S., Kim, M., Koh, W., Chang, J. W., Kim, H. J., Nakanishi, M., Barker, R. A., Na, D. L., Song, J.. iPSC Modeling of Presenilin1 Mutation in Alzheimer’s Disease with Cerebellar Ataxia. Experimental Neurobiology, 27, 27(5), 350-364, (2018)
Mae, S., Ryosaka, M., Toyoda, T., Matsuse, K., Oshima, Y., Tsujimoto, H., Okumura, S., Shibasaki, A., Osafune, K.. Generation of branching ureteric bud tissues from human pluripotent stem cells. Biochemical and biophysical research communications, 495(1), 954-961, (2018)
Musah, S., Dimitrakakis, N., Camacho, D. M., Church, G. M., Ingber, D. E.. Directed differentiation of human induced pluripotent stem cells into mature kidney podocytes and establishment of a lomerulus Chip. Nature protocols, 13(7), 1662, (2018)
Oshima, K., Saiki, N., Tanaka, M., Imamura, H., Niwa, A., Tanimura, A., Nagahashi, A., Hirayama, A., Okitac, K., Hotta, A., Kitayama, S., Osawa, M., Kaneko, S., Watanabe, A., Asaka, I., Fujibuchi, W., Imai, K., Yabe, H., Kamachi, Y., Hara, J., Kojima, S., Tomita, M., Soga, T., Noma, T., Nonoyama, S., Nakahata, T., Saito, MK.. Human AK2 links intracellular bioenergetic redistribution to the fate of hematopoietic progenitors. Biochemical and Biophysical Research Communications, 497(2), 719-725, (2018)
Saito, A., Ooki, A., Nakamura, T., Onodera, S., Hayashi, K., Hasegawa, D., Okudaira,T., Watanabe, K., Kato, H., Onda, T., Watanabe, A., Kosaki, K., Nishimura, K., Ohtaka, Manami., Nakanishi, M., Sakamoto, T., Yamaguchi, A., Sueishi, K., Azuma, T.. Targeted reversion of induced pluripotent stem cells from patients with human cleidocranial dysplasia improves bone regeneration in a rat calvarial bone defect model. Stem Cell Research & Therapy, 9(1), 12, (2018)
Sougawa, N., Miyagawa, S., Fukushima, S., Kawamura, A., Yokoyama, J., Ito, E., Harada, A., Okimoto, K., Mochisuki-Oda, N., Saito, A., Sawa, Y.. Immunologic targeting of CD30 eliminates tumourigenic human pluripotent stem cells, allowing safer clinical application of hiPSC-based cell therapy. Scientific Reports, 8(1), 3726, (2018)
Takayama, K., Hagihara, Y., Toba, Y., Sekiguchi, K., Sakurai, F., Mizuguchi, H.. Enrichment of high-functioning human iPS cell-derived hepatocyte-like cells for pharmaceutical research. Biomaterials, (2018)
Yamauchi, K., Li, J., Morikawa, K., Liu, L., Shirayoshi, Y., Nakatsuji, N., Elliott, A. D., Hisatome, I., Suemori, H..Isolation and characterization of ventricular-like cells derived from NKX2-5 eGFP/w and MLC2v mCherry/w double knock-in human pluripotent stem cells. Biochemical and Biophysical Research Communications, 495(1), 1278-1284, (2018)
Yasuda, S. Y., Ikeda, T., Shahsavarani, H., Yoshida, N., Nayer, B., Hino, M., Vartak-Sharma, N.,Suemori, H., Hasegawa, K.. Chemically defined and growth-factor-free culture system for the expansion and derivation of human pluripotent stem cells. Nature Biomedical Engineering, 2(3), 173, (2018)
Zhang, R. R., Koido, M., Tadokoro, T., Ouchi, R., Matsuno, T., Ueno, Y., Sekine, K., Takebe, T., Taniguchi, H.. Human iPSC-Derived Posterior Gut Progenitors Are Expandable and Capable of Forming Gut and Liver Organoids. Stem Cell Reports, 10(2), 1?14, (2018)
Camp, J. G., Sekine, K., Gerber, T., Loeffler-Wirth, H., Binder, H., Gac, M., Kanton, S., Kageyama, J., Damm, G., Seehofer, D., Belicova, L., Barsacchi, M., Barsacchi, R., Okuda, R., Yoshizawa, E., Kimura, M., Ayabe, H., Taniguchi, H., Takebe, T., & Belicova, L.. Multilineage communication regulates human liver bud development from pluripotency. Nature, 546, 533-538, (2017)
Goparaju, S. K., Kohda, K., Ibata, K., Soma, A., Nakatake, Y., Akiyama, T., Wakabayashi, S., Matsushita, M., Sakota, M., Kimura, H., Yuzaki, M., Shigeru B. H. Ko & Minoru S. H. Ko. . Rapid differentiation of human pluripotent stem cells into functional neurons by mRNAs encoding transcription factors. Scientific Reports, 7, 42367, (2017)
Hayashi, R., Ishikawa, Y., Katori, R., Sasamoto, Y., Taniwaki, Y., Takayanagi, Tsujikawa, M., Sekiguchi, K., Quantock, A. J., Nishida, K. . Coordinated generation of multiple ocular-like cell lineages and fabrication of functional corneal epithelial cell sheets from human iPS cells. Nature Protocols, 12(4), 683-696, (2017)
Hongo, A., Okumura, N., Nakahara, M., Kay, E. P., & Koizumi, N.. The Effect of a p38 Mitogen-Activated Protein Kinase Inhibitor on Cellular Senescence of Cultivated Human Corneal Endothelial CellsEffect of a p38 MAPK Inhibitor on Corneal Endothelial Cells. Investigative Ophthalmology & Visual Science, 58(9), 3325-3334, (2017)
Kikuchi, T., Morizane, A., Doi, D., Magotani, H., Onoe, H., Hayashi, T., Mizuma, H., Takara, S., Takahashi, R., Inoue, H., Morita, S., Yamamoto, M., Okita, K., Nakagawa, M., Parmar, M., Takahashi, J.. Human iPS cell-derived dopaminergic neurons function in a primate Parkinson’s disease model. Nature, 548, 592-596, (2017)
Kikuchi, T., Morizane, A., Okita, K., Nakagawa, M., Yamakado, H., Inoue, H., Takahashi, R., Takahashi, J. . Idiopathic Parkinson’s disease patient‐derived induced pluripotent stem cells function as midbrain dopaminergic neurons in rodent brains. Journal of Neuroscience Research, 95(9),1829-37, (2017)
Kojima, Y., Sasaki, K., Yokobayashi, S., Sakai, Y., Nakamura, T., Yabuta, Y., Nakaki, F., Nagaoka, S., Woltjen, K., Hotta, A., Yamamoto, T., Saitou, M.. Evolutionarily Distinctive Transcriptional and Signaling Programs Drive Human Germ Cell Lineage Specification from Pluripotent Stem Cells. Cell Stem Cell, 21(4), 517-532.e5, (2017)
Miyazaki, T., Isobe, T., Nakatsuji, N., & Suemori, H. . Efficient Adhesion Culture of Human Pluripotent Stem Cells Using Laminin Fragments in an Uncoated Manner. Scientific Reports, 7(41165), 1-8, (2017)
Morizane, A., Kikuchi, T., Hayashi, T., Mizuma, H., Takara, S., Doi, H., Mawatari, A., Glasser, M. F., Shiina, T., Ishigaki, H., Itoh, Y., Okita, K., Yamasaki, E., Doi, D., Onoe, H., Ogasawara, K., Yamanaka, S., and Takahashi, J. . MHC matching improves engraftment of iPSC-derived neurons in non-human primates. Nature Communications, 8(1), 385, (2017)
Musah, S., Mammoto, A., Ferrante, C. T., Jeanty, S.S., Hirano-Kobayashi, M., Mammoto, T., Roberts, K., Chung, S., Novak, R., Ingram, M., Fatanat-Didar, T., Koshy, S., Weaver, C. J., Church, M. G., Ingber, F. D. . Mature induced-pluripotent-stem-cell-derived human podocytes reconstitute kidney glomerular-capillary-wall function on a chip. Nature Biomedical Engineering, 1 (0069), (2017)
Polisetti, N., Sorokin, L., Okumura, N., Koizumi, N., Kinoshita, S., Kruse, F. E., and Schlotzer-Schrehardt, U. Laminin-511 and-521-based matrices for efficient ex vivo-expansion of human limbal epithelial progenitor cells. Scientific Reports, 7, 5152, (2017)
Sato-Nishiuchi, R., Li, S., Ebisu, F., Sekiguchi, K.. Recombinant laminin fragments endowed with collagen-binding activity: A tool for conferring laminin-like cell-adhesive activity to collagen matrices. Matrix Biology, (2017)
Sekine, S. I., Kondo, T., Murakami, N., Imamura, K., Enami, T., Shibukawa, R., Tsukita, K., Funayama, M., Inden, M., Kurita, H., Hozumi, I., Inoue, H.. Induced pluripotent stem cells derived from a patient with familial idiopathic basal ganglia calcification (IBGC) caused by a variant in SLC20A2 gene. Stem Cell Research, (2017)
Sougawa, N., Miyagawa, S., Fukushima, S., Saito, A., Yokoyama, J., Kitahara, M., Harada, A., Sato-Nishiuchi, R., Sekiguchi, K., Sawa, Y.. Novel Stem Cell Niches Laminin 511 Promotes Functional Angiogenesis Through Enhanced Stem Cell Homing by Modulating” Stem Cell Beds” in the Failed Heart.Circulation, 136(1), A15587, (2017)
Taguchi, A., & Nishinakamura, R.. Higher-Order Kidney Organogenesis from Pluripotent Stem Cells. Cell Stem Cell, 21. (2017)
Takebe, T., Sekine, K., Kimura, M., Yoshizawa, E., Ayano, S., Koido, M., Funayama, S., Nakanishi, N., Hisai, T., Kobayashi, T., Kasai, T., Kitada, R., Mori, A., Ayabe, H., Ejiri, Y., Amimoto, N., Yamazaki, Y., Ogawa, S., Ishikawa, M., Kiyota, Y., Sato, Y., Nozawa, K., Okamoto, S., Ueno, Y., Kasai, T.. Massive and Reproducible Production of Liver Buds Entirely from Human Pluripotent Stem Cells. Cell Reports, 21(10), 2661-2670, (2017)
Takizawa, M., Arimori, T., Taniguchi, Y., Kitago, Y., Yamashita, E., Takagi, J., Sekiguchi, K.. Mechanistic basis for the recognition of laminin-511 by α6β1 integrin. Science Advances, 3(9), e1701497, (2017)
Tan, G. W., Kondo, T., Murakami, N., Imamura, K., Enami, T., Tsukita, K., Shibukawa, R., Funayama, M., Matsumoto, R., Ikeda, I., Takahashi, R., Inoue, H.. Induced pluripotent stem cells derived from an autosomal dominant lateral temporal epilepsy (ADLTE) patient carrying S473L mutation in leucine-rich glioma inactivated 1 (LGI1). Stem Cell Research, (2017)
Taniguchi, Y., Li, S., Takizawa, M., Oonishi, E., Toga, J., Yagi, E., & Sekiguchi, K. Probing the acidic residue within the integrin binding site of laminin-511 that interacts with the metal ion-dependent adhesion site of α6β1 integrin. Biochemical and Biophysical Research Communications, 487(3), 525-531, (2017)
Uchimura, T., Otomo, J., Sato, M., Sakurai, H.. A human iPS cell myogenic differentiation system permitting high-throughput drug screening. Stem cell research, 25, 98-106, (2017)
Hayashi, R., Ishikawa, Y., Sasamoto, Y., Katori, R., Nomura, N., Ichikawa, T., Araki, S., Soma, T., Kawasaki, S., Sekiguchi, K., Tsujikawa, M., Nishida, K., & Quantock, A. J.. Co-ordinated ocular development from human iPS cells and recovery of corneal function. Nature, 531(7594), 376-380, (2016)
Kawamura, T., Miyagawa, S., Fukushima, S., Maeda, A., Kashiyama, N., Kawamura, A., Miki, K., Okita, K., Yoshida, Y., Shiina, T., Ogasawara, K., Miyagawa, S., Toda, K., Okuyama, H., Sawa, Y.. Cardiomyocytes derived from MHC-homozygous induced pluripotent stem cells exhibit reduced allogeneic immunogenicity in MHC-matched non-human primates. Stem cell reports, 6(3), 312-320, (2016).
Matsuno, K., Mae, S. I., Okada, C., Nakamura, M., Watanabe, A., Toyoda, T., Uchida, E., Osafune, K.. Redefining definitive endoderm subtypes by robust induction of human induced pluripotent stem cells. Differentiation; research in biological diversity, (2016)
Nishimura, K., Doi, D., Samata, B., Murayama, S., Tahara, T., Onoe, H., & Takahashi, J.. Estradiol Facilitates Functional Integration of iPSC-Derived Dopaminergic Neurons into Striatal Neuronal Circuits via Activation of Integrin α5β1. Stem cell reports, 6(4), 511-524, (2016)
Samata, B., Doi, D., Nishimura, K., Kikuchi, T., Watanabe, A., Sakamoto, Y., Kakuta, J., Ono, Y., & Takahashi, J.. Purification of functional human ES and iPSC-derived midbrain dopaminergic progenitors using LRTM1. Nature Communications, 7(13097), 1-11, (2016)
Takayama, K., Mitani, S., Nagamoto, Y., Sakurai, F., Tachibana, M., Taniguchi, Y., Sekiguchi, K., Mizuguchi, H.. Laminin 411 and 511 promote the cholangiocyte differentiation of human induced pluripotent stem cells. Biochemical and biophysical research communications, 474(1), 91-96, (2016)
Tanigawa, S., Taguchi, A., Sharma, N., Perantoni, A. O., & Nishinakamura, R.. Selective in vitro propagation of nephron progenitors derived from embryos and pluripotent stem cells. Cell reports, 15(4), 801-813, (2016)
Burridge, P. W., Matsa, E., Shukla, P., Lin, Z. C., Churko, J. M., Ebert, A. D., Lan, F., Diecke, S., Huber, B., Mordwinkin, N. M., Plews, J. R., Abilez, O. J., Cui, B., Gold, J. D., & Wu, J. C. . Chemically defined generation of human cardiomyocytes. Nature methods, 11(8), 855-860, (2014)
Doi, D., Samata, B., Katsukawa, M., Kikuchi, T., Morizane, A., Ono, Y., Sekiguchi, K., Nakagawa, M., Parmar, M., Takahashi, J.. Isolation of human induced pluripotent stem cell-derived dopaminergic progenitors by cell sorting for successful transplantation. Stem cell reports, 2(3), 337-350, (2014)
Fukuta, M., Nakai, Y., Kirino, K., Nakagawa, M., Sekiguchi, K., Nagata, S., Matsumoto, Y., Yamamoto, T., Umeda, K., Heike, T., Okumura, N., Koizumi, N., Sato, T., Nakahata, T., Saito, M., Otsuka, T., Kinoshita, S., Ueno, M., Ikeya, M., Toguchida, J. . Derivation of mesenchymal stromal cells from pluripotent stem cells through a neural crest lineage using small molecule compounds with defined media. PloS one, 9(12), e112291, (2014)
Ido, H., Nakamura, A., Kobayashi, R., Ito, S., Li, S., Futaki, S., & Sekiguchi, K. . The requirement of the glutamic acid residue at the third position from the carboxyl termini of the laminin γ chains in integrin binding by laminins. Journal of Biological Chemistry, 282(15), 11144-11154, (2007)
Miyazaki, T., Futaki, S., Suemori, H., Taniguchi, Y., Yamada, M., Kawasaki, M., Hayashi, M., Kumagai, H., Nakatsuji, N., Sekiguchi, K., & Kawase, E. . Laminin E8 fragment support efficient adhesion and expansion of dissociated human pluripotent stem cells. Nature communications, 3(1236), 1-10, (2012)
Nakagawa, M., Taniguchi, Y., Senda, S., Takizawa, N., Ichisaka, T., Asano, K., Morizane, A., Doi, D., Takahashi, J., Nishizawa, M., Yoshida, Y., Toyoda, T., Osafune, K., Sekiguchi, K., & Yamanaka, S. . A novel efficient feeder-free culture system for the derivation of human induced pluripotent stem cells. Scientific reports, 4(3594), 1-7, (2014)
Okumura, N., Kakutani, K., Numata, R., Nakahara, M., Schlotzer-Schrehardt, U., Kruse, F., Kinoshita. K., Koizumi, N.. Laminin-511 and-521 Enable Efficient In Vitro Expansion of Human Corneal Endothelial CellsLaminin-511 and-521 Enable Expansion of HCECs. Investigative ophthalmology & visual science, 56(5), 2933-2942, (2015)
Sasaki, K., Yokobayashi, S., Nakamura, T., Okamoto, I., Yabuta, Y., Kurimoto, K., Ohta, H., Moritoki, Y., Iwatani, C., Tsuchiya, H., Nakamura, S., Sekiguchi, K., Sakuma, T., Yamamoto, T., Mori, T., Woltjen, K., Nakagawa, M., Yamamoto, T., Takahashi, K., Yamanaka, S., Saitou, M.. Robust in vitro induction of human germ cell fate from pluripotent stem cells. Cell stem cell, 17(2), 178-194, (2015)
Takashima, Y., Guo, G., Loos, R., Nichols, J., Ficz, G., Krueger, F., Oxley, D., Santos, F., Clarke, J., Mansfield, W., Reik, W., Bertone, P., Smith, A.. Resetting transcription factor control circuitry toward ground-state pluripotency in human. Cell, 158(6), 1254-1269, (2014)
Taniguchi, Y., Ido, H., Sanzen, N., Hayashi, M., Sato-Nishiuchi, R., Futaki, S., & Sekiguchi, K. . The C-terminal region of laminin β chains modulates the integrin binding affinities of laminins. Journal of Biological Chemistry, 284(12), 7820-7831, (2009)
Other Documents
- Protocol iMatrix-511
- Protocol iMatrix-511-Silk
- Protocol Easy iMatrix-511/iMatrix-511-Silk
- MSDS iMatrix-511
- MSDS iMatrix-511-Silk
- MSDS Easy-iMatrix-511
- MSDS Easy-iMatrix-511-Silk
- ECM Pre-mix method
- Detachment Method using EDTA for human iPS cells
Related Products
FOR RESEARCH USE ONLY, NOT FOR USE IN DIAGNOSTIC PROCEDURES